The RUS platform uses advanced imaging technology to transform CT scans into detailed 3D models, enabling precise surgical planning and intraoperative navigation. Designed for gastric and renal procedures, RUS GA and RUS NE support comprehensive preoperative planning, while RUS CREATOR generates patient-specific 3D models to support improved surgical outcomes.
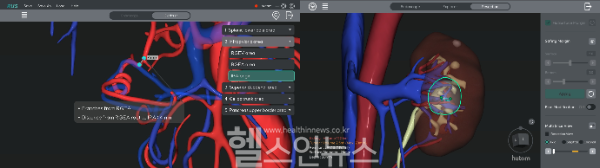
Already widely adopted in South Korea’s major tertiary hospitals, the RUS series has recently expanded into thoracic surgery. The newly developed RUS LUNG solution enhances the safety and precision of lung resections by accurately visualizing complex pulmonary vasculature and segmental anatomy.
Hutom’s global expansion accelerated last year when its RUS Stomach solution received 510(k) clearance from the U.S. Food and Drug Administration (FDA). The company also secured a provisional evaluation under South Korea’s New Medical Technology Assessment program. The Hong Kong registration is expected to strengthen Hutom’s entry into the broader Asian market.
"Approval in Hong Kong opens important pathways for us in Asia," said Woojin Hyung, CEO of Hutom. "Our AI-powered surgical navigation solutions are designed to improve patient outcomes in hospitals worldwide."
Kim Kuk Ju, HEALTH IN NEWS TEAM
press@hinews.co.kr


